In the pharmaceutical industry, product quality is non-negotiable. Every tablet, capsule, or injectable medicine must deliver the correct amount of active ingredients to ensure patient safety and therapeutic effectiveness. This is where assay testing plays a vital role.
Assay testing is one of the most critical quality control tests performed during drug development and commercial manufacturing. It determines the exact quantity of active pharmaceutical ingredients (API) present in a drug product and confirms compliance with regulatory and pharmacopeial requirements.
What Is Assay Testing?
Assay testing is an analytical procedure used to measure the strength or potency of a pharmaceutical product. It verifies whether the drug contains the correct amount of API as stated on its label.
Common analytical techniques used for assay testing include:
- High-Performance Liquid Chromatography (HPLC)
- UV-Visible Spectroscopy
- Titration methods
- Gas Chromatography (GC)
These methods are selected based on the product type, formulation, and regulatory expectations.
Why Assay Testing Matters
- Ensuring Patient Safety
Accurate dosage is essential for patient health and treatment outcomes.
- Under-dosed medicines may fail to deliver therapeutic benefit.
- Over-dosed medicines can cause toxicity and serious adverse effects.
Assay testing ensures patients receive the intended dose every time, reducing risk and maximizing therapeutic effectiveness.
- Guaranteeing Product Quality and Consistency
Pharmaceutical manufacturers must demonstrate batch-to-batch consistency. Assay testing confirms that every production lot meets the same potency specifications, ensuring uniform product performance.
This consistency is critical for:
- Reliable clinical outcomes
- Brand reputation
- Long-term patient and provider trust
- Meeting Regulatory Requirements
Global regulatory agencies such as the FDA, WHO etc. require assay testing as part of Good Manufacturing Practices (GMP).
Drug products that fail assay specifications may be:
- Rejected during release testing
- Subject to recalls
- Flagged for regulatory enforcement actions
Routine assay testing ensures ongoing compliance with regulatory expectations.
- Verifying Label Claims
Assay testing confirms that the labeled strength of a pharmaceutical product accurately reflects its contents. This protects healthcare professionals and patients from misleading information and supports transparency and regulatory compliance in labeling.
- Supporting Stability and Shelf-Life Studies
During stability studies, assay results are used to evaluate how API potency changes over time. This data is essential for:
- Establishing expiration dates
- Confirming product potency throughout shelf life
- Ensuring product effectiveness through the end of use
- Detecting Manufacturing Errors Early
Assay testing can uncover potential manufacturing issues, including:
- Incorrect API weighing
- Inadequate blending or mixing
- API degradation during processing
Early detection prevents non-conforming products from reaching the market, saving time, cost, and protecting patient safety.
- Validating Manufacturing Processes
Consistent assay results provide evidence that:
- The formulation is correctly designed
- The manufacturing process is robust and controlled
- Quality systems are functioning as intended
This data supports process validation, continuous improvement, and lifecycle management.
Conclusion
Assay testing is a cornerstone of pharmaceutical quality control. It ensures that medicines are safe, effective, consistent, and compliant with regulatory standards. Without accurate assay testing, there is no reliable way to guarantee product potency or protect patient safety.
In an industry where precision matters, assay testing is not just a regulatory requirement. It is a fundamental responsibility.
How RMA Can Support Your Assay Testing Needs
The RMA team brings years of experience performing assay testing for raw materials and finished pharmaceutical products as per various compendial methods such as USP, EP, JP, JPE etc. across development and commercial manufacturing stages.
All testing is performed with strict adherence to cGMP requirements in an FDA-registered and FDA-audited laboratory, ensuring data integrity, regulatory compliance, and confidence in every result.
Whether you are supporting product development, batch release, stability studies, or regulatory submissions, RMA is here to help.
Contact us today: engage@rawmaterialanalytical.com
Call us: 833-928-8333
Recent Blogs and News
Why Raw Material and Finished Product Testing Is Mandatory in the Pharmaceutical Industry
Raw material and finished product testing isn't optional in pharma; it's federal law. Learn why the FDA mandates these crucial tests for patient safety and cGMP compliance
Why Assay Testing Is So Important in the Pharmaceutical Industry
Why is assay testing critical in pharma? It verifies the exact strength of active ingredients (API) to ensure accurate dosages, product consistency, and patient safety.
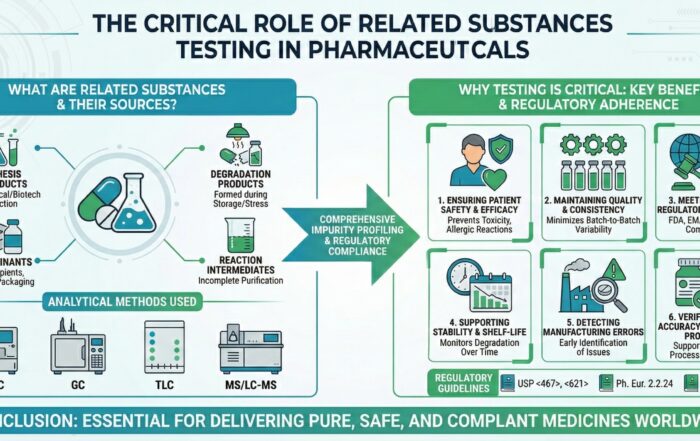
What Are Related Substances?
Discover why related substances testing is critical in pharmaceuticals. Learn about impurity sources, FDA/ICH regulatory compliance, and ensuring patient safety.