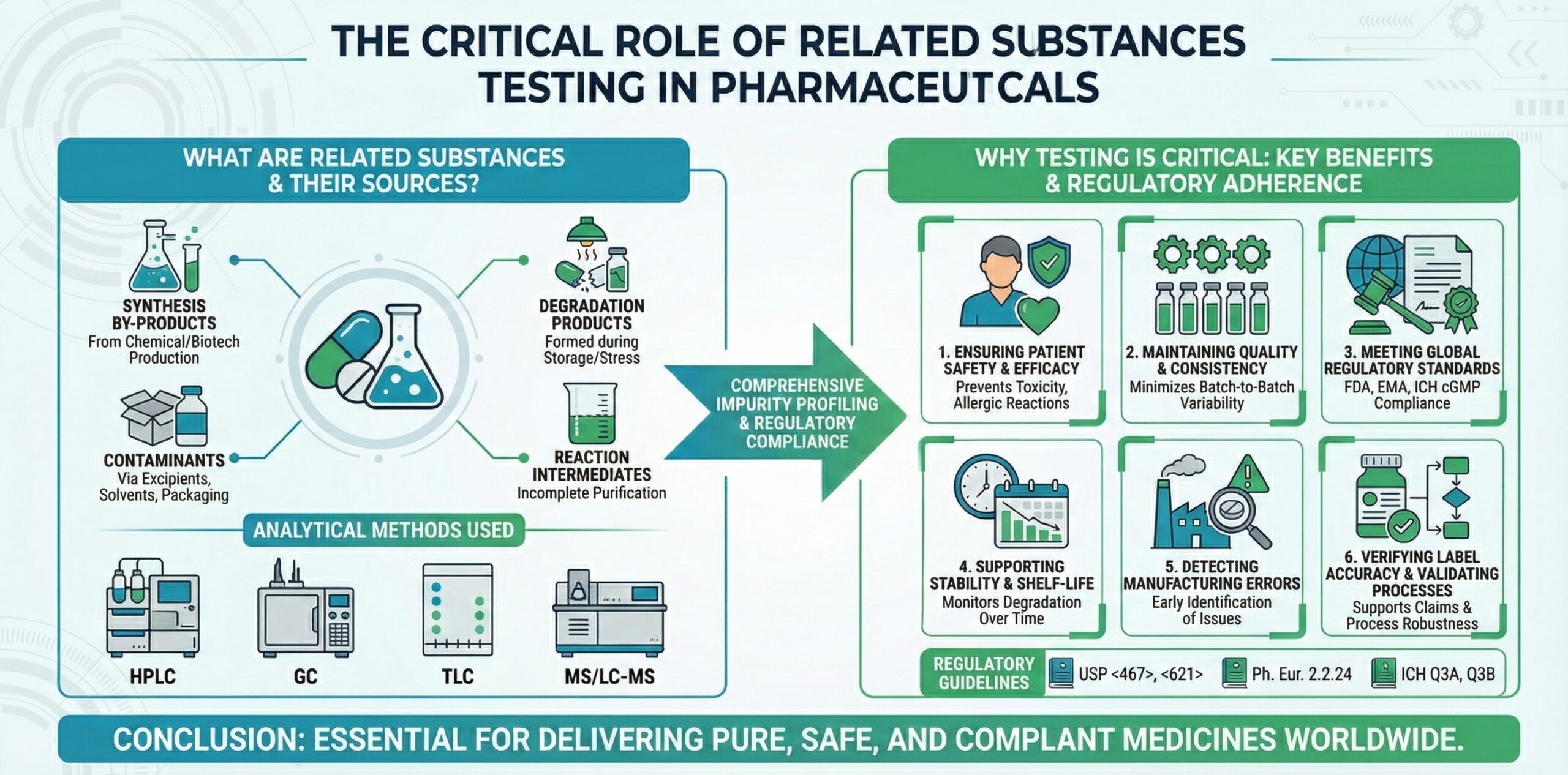
Related substances include any chemical entity present in a drug substance or drug product that is not the intended API. These substances can arise from:
- Synthesis by-products in the chemical or biotechnological production of the API
- Degradation products formed during processing, storage, or under stress conditions
- Contaminants introduced via excipients, solvents, or packaging materials
- Reaction intermediates from incomplete purification
While some related substances may be harmless, others can be toxic, reduce drug efficacy, or affect product stability. Consequently, regulatory authorities require their detection, identification, and quantification within defined acceptable limits.
Why Related Substances Testing Is Critical
- Ensuring Patient Safety
Even minute amounts of impurities can compromise drug safety. Toxic degradation products or reactive by-products may lead to:
- Allergic reactions or hypersensitivity
- Organ toxicity or systemic effects
- Reduced therapeutic efficacy
Related substances testing ensures that the final drug product is safe for patients and meets stringent pharmacopeial purity requirements.
- Maintaining Product Quality and Consistency
Consistency is key in pharmaceutical manufacturing. Related substances testing ensures that each batch of a drug substance or product maintains the same purity profile, minimizing batch-to-batch variability. This is vital for:
- Predictable clinical outcomes
- Reliable pharmacological performance
- Maintaining brand reputation and patient trust
- Compliance with Global Regulatory Standards
Regulatory agencies such as the FDA, EMA, WHO etc. mandate related substances testing as part of current Good Manufacturing Practices (cGMP). Failure to meet pharmacopeial impurity limits can result in:
- Batch rejection during release testing
- Market recalls or withdrawals
- Regulatory inspections and warning letters
Routine testing ensures products consistently meet global standards and avoids costly compliance issues.
- Supporting Stability and Shelf-Life Studies
Pharmaceutical products are subject to chemical, physical, and environmental stresses during storage. Related substances testing is critical for stability studies, helping to:
- Monitor degradation pathways of the API
- Determine how impurities form over time
- Establish accurate expiration dates
- Optimize packaging and storage conditions
By understanding the stability profile, manufacturers can ensure drugs remain potent and safe throughout their shelf life.
- Detecting Manufacturing and Process Errors
Manufacturing a pharmaceutical product involves multiple complex steps. Related substances testing can reveal:
- Incorrect raw material or API handling
- Incomplete purification of intermediates
- Inadequate mixing or blending
- Degradation due to heat, moisture, or light exposure
Early detection of such issues prevents substandard products from reaching patients, reducing regulatory and financial risks.
- Verifying Label Accuracy and Regulatory Claims
Regulators and healthcare providers rely on accurate labeling of drug products. Related substances testing ensures that:
- Impurities do not exceed specified limits
- Product labels reflect accurate information about strength, purity, and composition
- Regulatory dossiers and submissions are fully supported with reliable analytical data
This transparency builds trust among healthcare professionals and patients.
- Validating Manufacturing Processes
High-quality analytical testing supports process validation. By consistently monitoring related substances:
- The robustness of synthesis and formulation processes is demonstrated
- Quality control measures can be optimized
- Continuous improvement and risk mitigation strategies are supported
Regulators view comprehensive impurity profiling as a critical indicator of GMP compliance and process control.
Analytical Methods for Related Substances Testing
A variety of validated analytical techniques are used to detect and quantify impurities:
- High-Performance Liquid Chromatography (HPLC) – The most widely used method for identifying and quantifying impurities in drug substances and products.
- Gas Chromatography (GC) – Effective for volatile or thermally stable substances.
- Thin Layer Chromatography (TLC) – Useful for qualitative detection of minor impurities.
- UV-Visible Spectroscopy – Applied for preliminary or supporting analysis of chromophoric impurities.
- Mass Spectrometry (MS) and LC-MS/MS – Provide structural information and identification of unknown or complex impurities.
Selection of the method depends on the drug formulation, regulatory requirements, and the nature of potential impurities.
Regulatory Standards and Guidelines
Pharmacopeial and regulatory guidance provide specific limits and methodologies for related substances testing:
- USP <467> – Residual solvents and impurities
- USP <621> – Chromatography-based analysis
- Ph. Eur. 2.2.24 – Related substances testing using HPLC
- ICH Q3A and Q3B – Impurities in new drug substances and drug products
Adhering to these standards ensures regulatory acceptance and patient safety.
How RMA Can Support Your Related Substances Testing Needs
The RMA team brings extensive expertise in performing related substances testing for both drug substances and finished products. We work with clients to develop fit-for-purpose analytical strategies that meet USP, Ph. Eur, ICH, and global regulatory expectations.
At RMA:
- All testing is performed in a cGMP-compliant, FDA-registered, and FDA-audited laboratory
- We provide method development, validation, and routine analysis for impurity profiling
- Testing supports product development, batch release, stability studies, and regulatory submissions
- Reports are comprehensive, defensible, and aligned with regulatory requirements
Whether you are qualifying new suppliers, troubleshooting manufacturing processes, or preparing for regulatory filings, RMA is ready to ensure your products meet the highest standards of safety and quality.
Contact us today: engage@rawmaterialanalytical.com
Call us: 833-928-8333
Recent Blogs and News
Why Raw Material and Finished Product Testing Is Mandatory in the Pharmaceutical Industry
Raw material and finished product testing isn't optional in pharma; it's federal law. Learn why the FDA mandates these crucial tests for patient safety and cGMP compliance
Why Assay Testing Is So Important in the Pharmaceutical Industry
Why is assay testing critical in pharma? It verifies the exact strength of active ingredients (API) to ensure accurate dosages, product consistency, and patient safety.
What Are Related Substances?
Discover why related substances testing is critical in pharmaceuticals. Learn about impurity sources, FDA/ICH regulatory compliance, and ensuring patient safety.